Protocols for SNP Markers
PCR
The PCR setup to for all SNP markers is as follows:
| Reagent | Stock conc. | Vol. (ul) per rxn | rxn conc. |
| Promega Buffer* | 10X | 1.5 | 1X |
| MgCl2 | 25mM | 1.8 | 3mM |
| dNTPs | 1.25mM | 2.4 | 0.2mM |
| Template DNA | 10ng/ul | 1.5 | 15ng (1ng/l) |
| Primer | 5uM | 1 | 0.3uM |
| Taq | ? | 0.4 | ~1 U |
| H2O | 6.4 | ||
| Total | 15 |
*Progema PCR buffer w/o MgCl2
To download an excel sheet to give calculations for Master Mix, click here.
PCR reaction conditions were as follows:
Initial melting of
94oC for 5min
35 cycles of:
94oC for 30sec
XoC for 45sec
72oC for 1min
Then final extension of
72oC for 6min
Where XoC is the annealing temperature depending on the marker.
Gel Electrophoresis
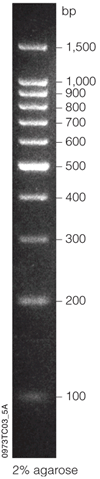
All SNP markers from this project can be resolved on 1.6% agarose gels run in Staub lab gel boxes (OWL Gator A3) at 180V for 2 hours. Ladder used was Promega 100 bp ladder (cat#G2101) (see photo below)

This page last updated: Friday January 20, 2006 16:59